Governments meet the people, but against pharma
In 2017-2018, the US government actively discussed
In Russia after unpopular pension reformthe government will seek to compensate for public discontent with socially-oriented projects. With high probability in 2019, the development of price regulation for many socially important drugs will begin, given the speed at which regulatory decisions are made, it is possible that by 2019–2020 such regulatory acts will be adopted.
This state of affairs is fraught with financial losses.for a number of companies of industry value. These manufacturers certainly predict a similar development of the situation and reduce costs. As in other situations, with a decrease in the expenditure part of pharmaceutical manufacturers, first of all, budgets for new development and marketing will fall under the cuts.
Mobile applications for the needs of the pharmaceutical industry
A particular influence on pharma is the widespread use of fitness applications and peripheral monitoring devices.
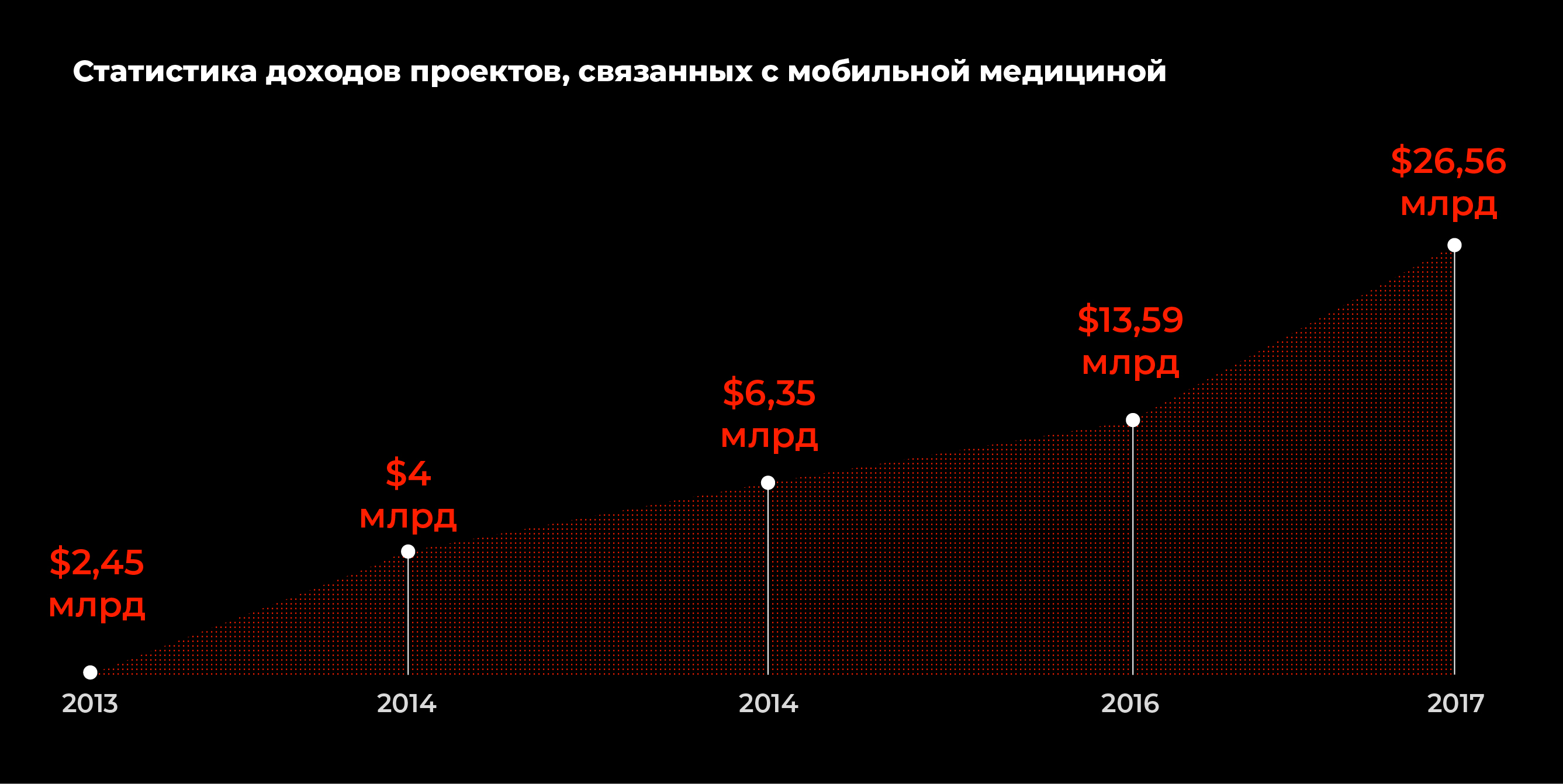
Obviously, pharmacological costs are rising.According to statistics, in 2018, the number of pharmaceutical advertising companies in digital marketing and advertising amounted to $ 2.84 billion. Volumes will increase regularly. Thus, by the number of downloads on Google Play, the average application for doctors, developed by the pharmaceutical manufacturer, is downloaded about 3-5 thousand times, and third-party medical applications containing pharmaceutical advertising, about 4-5 million times.
Such a pronounced global trend can not benot seen in Russia, but like most digital innovations in marketing, it will be perceived by the market with a lag of 1-1.5 years. It is also known that in Russia several own projects are being developed on medical applications that plan to monetize through advertising, which is guaranteed to attract pharmaceutical manufacturers as advertisers.
Predictive analytics for accurate medicine
In the West, in particular, in the EU is actively developingpredictive analytics, in the development of which participants of the pharmacological market and the biotechnology industry play a special role. All Western companies are involved in the collection and analytical processing of statistical data, which conduct clinical trials and bring new drugs to the market.
Sidebar
The trend is ingrained in the EU and the USA, to the presentCurrently, prognostic analytics techniques are an integral part of Western medicine and the pharmacological industry. Many experts write about the prospects in this area, in particular, Arun Bondali.
Ignore predictive featuresAnalytics of statistical data in Russia is extremely difficult, first of all, due to the rather large amount of such data and their high diagnostic and clinical value. The scale of possible projects in this area will affect both pharmacological companies and research circles of medical universities in 2019, just as it did in the United States and Europe earlier.
Centralization of development: fewer patents, more generics
Trend for the use of third-party developmentThere is a long time and not only in the United States. The existing position in the global drug market, as well as the regulatory features of the pharmaceutical industry, are provoking the increasing centralization and globalization of new developments.
Modern tough requirements thatare presented for testing and clinical trials of new drugs, make it impossible to develop for small and medium-sized companies, and also significantly increase the period of development of new drugs. The reduction in development costs described above also revives this rather long-running trend.
Pharmaceutical Research and Manufacturers DataOf America (PhRMA) indicate that the production of a new drug costs about $ 900 million (30 years ago this amount did not exceed $ 300 million). According to their own information, the average development time ranges from 10–15 years. Biotech drugs require even more money and time, the costs of their development often exceed $ 1 billion, and the terms can be up to 20 years.
It is also known that to become profitable and, byat least, only one-third of the drugs on the market are able to reach zero in the United States. Despite this, across the country, every dollar spent on health care, including pharmacological and technological development, reduces the cost of situations related to disability by $ 3. Such statistics is typical for the United States for several decades.
Obviously, it is more profitable for Russian companies.produce generics, which they have been successfully engaged in over the past ten years. In the future, the number of generics of domestic production will only grow - this is the only fast and cheap way to meet the growing needs of the domestic market, in which brand-name drugs look not very attractive due to the high cost.
Trend to develop unprofitable in the shortover the long term, but the use of proprietary tools that will be subsidized by the state for the sake of social effect, can also take root on Russian soil. The key to this trend will be the question of the significance of the social effect. The higher the social effect and economic benefits from its use, the higher the level of subsidies may be.
Active use of big data and IoT
In the West, this trend began in 2015-2016years, when the largest pharmacological companies began to use data center services. In fact, this trend stems from the two previous ones, which involve the use of powerful data centers for data collection and processing, as well as the use of various peripheral devices to obtain meaningful information about potential drug users.
Global Data experts expect market volumesIoT software and services in the pharmaceutical industry will grow to $ 2.4 billion by 2020. The growth trend implies active development of big data and investments in the corresponding infrastructure. Back in 2013, the long-term forecast by James Cattell of McKinsey reported that big data could increase the total cost of the pharmaceutical industry by $ 100 billion, solely by improving research and development.
Russian manufacturers also will not stay into the side. The rapid construction of large data centers (for example, Mendeleev), obviously, is being carried out with a view to meeting the needs of the pharmacological market. Thematic events are taking place, for example, in Moscow, the conference “Greater Data and Artificial Intelligence in Pharma” was held. The situation is similar with IoT, which is rated as one of the leading directions in the Russian national IT segment.
The most striking example of using IoT onThe West is Amazon’s experience and the medical and pharmacological use of the AWS platform. The cloud array helps to simplify the introduction of technological innovations in the pharmacological industry, simplifies the application and integration of high-performance computing and machine learning for the development of pharma. The company's plans include a new service that will simplify work with clinical data registration systems, prescription of drugs, as well as the choice of drugs at an optimal cost.
It is assumed that the new service Amazon willsuggest how best to treat patients and save on drugs. The company plans to include in the service the recognition of medical records and the ability to make voice recommendations. The company even stated that “medical” handwriting would not be a problem in recognition.
A similar project with a clear focus onpharmacological market is already developing in Russia. The participants of the information and educational project Pharmedu set the task of creating an integrated set of educational opportunities and a knowledge base. And both for the professional community (pharmacists, pharmacists and others), and for pharmaceutical products consumers. For the latter, a sub-service “Pharmacy Kiosk” is being created within the project. As planned, it will allow the pharmacy buyer to prepare in advance for the visit and make an informed, competent choice in favor of the drug.
The relevance of such services is high becausethe consumer has to constantly compare his knowledge with the recommendations of doctors and pharmacists. It is known that such recommendations are sometimes redundant or not entirely accurate. This often occurs as a result of collusion between the doctor and pharmaceutical companies or aggressive commercial propaganda, which, regularly repeated, actually deprive patients of choice and further increase the need for neutral referral services.
More drug development for Alzheimer's
The trend is associated with the completion of the study, whichwas published in December 2018 by American scientists from the Southwest Medical Center of the University of Texas, the University of Delaware and the University of Washington at St. Louis. The study sheds light on the mechanisms of Alzheimer's disease. In the US, pharmaceutical manufacturers are expected to start developing drug products in the near future based on the research results.
Surely after a while the trend will come inRussia There are several possible options. The first is the creation and production of our own drugs, as was the case with some drugs against cancer and cardiovascular diseases, the creation of which was associated with basic research on the causes and pathogenesis of diseases. The second is the production of generics after the development and testing of the drug in the West.
Reducing the pool of biotech developers
The trend of biotech development affectsworld pharmaceutical industry last 15 years. Personalization of bioprocessing, increasing the effectiveness of drugs, unfortunately, can maintain profitability only in the medium term. The interdependence and integration of processes in these industries is very high. On this basis, we can say that the processes associated with the high cost of developing and launching new drugs to the market fully relate to the biotech industry.
Sidebar
Reducing the number of biotechdevelopers are expected in the near future, many experts predicted the beginning of this process last year and attributed it just to a slow payback and lower profitability in this area. These processes are caused by increased requirements for the quality of drugs and an attempt to prevent the market from being filled with “suboptimal” drugs. The scruples of large companies and the pressure of regulatory structures are forcing small firms and poorly funded start-ups to leave the market.
For Russia, the development of the described trend is possible with a tightening of regulatory control, as well as an increase in the cost and requirements for clinical research.
Obviously, not all players will be able to withstand quite severe market conditions and regulation. That is why the Western trend may well affect the realities of the domestic pharmaceutical market.
Of course, these are not all Western trends thatwill appear in the Russian pharmaceutical industry, but the most significant and noticeable. Obviously, some of the trends are clearly interconnected, and the manifestation of some will naturally draw the others along. But most trends will allow to create better and more effective drugs, as well as predictably avoid the complex consequences of the costs of government regulation.