One of the main reasons for the poor performance of some batteries is that the lithium they contain
Researchers from the University of TechnologyZhejiang in China and Argonne National Laboratory in the United States have developed a strategy for the recovery of inactive lithium in anodes. The new strategy is based on a chemical reaction known as the redox potential of iodine.
Solid electrolyte interfacial (SEI) isa layer that forms on the anode of lithium-ion batteries during the first few charging cycles. The passivation layer plays a critical role in battery performance, stability and safety.
In a typical lithium-ion battery cell withWith a conventional graphite anode, the solid electrolyte interfacial (SEI) phase usually consists of LiF in combination with Li₂CO₃, alkyl carbonate and other substances. Recent research has shown that, in lithium metal anode batteries, SEI is mainly composed of Li₂O rather than LiF. In these batteries, changing the volume of the Li-coating can compromise the mechanical integrity and passivation role of the Li SEO-based SEI. This, in turn, can lead to the formation of "dead lithium".
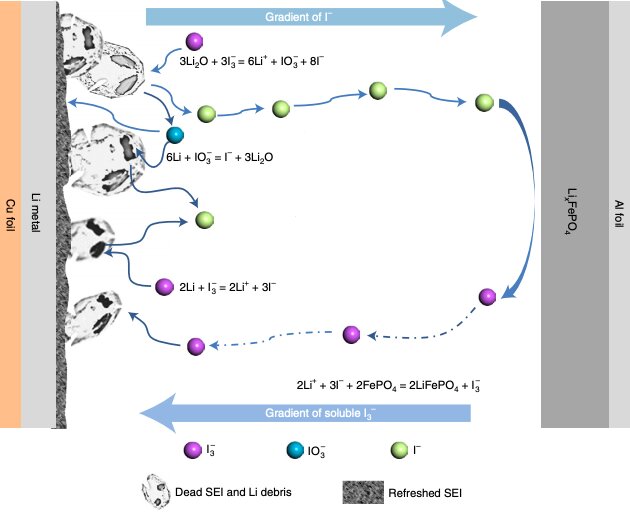 Figure summarizing the Li recovery strategy developed by the researchers. Credit: Jin et al. (Energy of Nature).
Figure summarizing the Li recovery strategy developed by the researchers. Credit: Jin et al. (Energy of Nature).
After conducting a study, scientists found out what exactlyLi loss in SEI and dead lithium particles are the main reasons for the performance degradation often seen in metal lithium batteries. This observation inspired them to develop a method for the reduction of dead lithium using the redox chemical reaction of iodine.
Scientists were able to create a fully chargedbattery cell with very low lithium content in the anode. This cell had a lifespan of 1000 cycles and achieved a Columbian system's high efficiency of 99.9%.
Read more
The first accurate map of the world was created. What's wrong with everyone else?
See streams of Martian dust from the Ingenuity helicopter
Ingenuity helicopter successfully takes off on Mars
A passivation layer is a film on the surface of a metal that protects the metal from corrosion under conditions where the metal is thermodynamically reactive.It is caused by the formation of protective surface compounds during the interaction of the metal with the components of the medium.In terms of composition, passivation layers are divided into oxide and salt, layers of more complex composition are possible.